Hybrid Membrane Captures Carbon Emissions
Engineering360 News Desk | March 30, 2016Part polymer and part metal-organic framework (MOF) -- a porous three-dimensional crystal -- a membrane could lead to more efficient ways of separating carbon dioxide from power plant exhaust. The highly permeable carbon capture material developed at U.S. Department of Energy’s Lawrence Berkeley National Laboratory (Berkeley Lab) features a large internal surface area that can absorb large amounts of molecules.
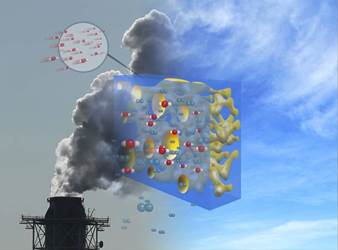 Exhaust contacts a hybrid membrane recently developed at Berkeley Lab.The hybrid membrane presents two channels for carbon dioxide molecule transport. Molecules can travel through the polymer component of the membrane, as in conventional gas-separation membranes. Or molecules can flow through “carbon dioxide highways” created by adjacent MOFs (see video).
Exhaust contacts a hybrid membrane recently developed at Berkeley Lab.The hybrid membrane presents two channels for carbon dioxide molecule transport. Molecules can travel through the polymer component of the membrane, as in conventional gas-separation membranes. Or molecules can flow through “carbon dioxide highways” created by adjacent MOFs (see video).
Initial tests show this two-route approach makes the hybrid membrane eight times more carbon dioxide permeable than membranes composed only of the polymer. Boosting carbon dioxide permeability is a goal in efforts to develop carbon capture materials that are energy efficient and cost competitive.
Polymer membranes often are preferred as a more energy efficient alternative to widely used amine adsorption technology. These membranes are relatively inexpensive and easy to work with, but current commercial membranes have low carbon dioxide permeability. To overcome this, scientists have developed hybrid membranes that are part polymer and part MOF. These hybrids harness the carbon dioxide selectivity of MOFs while maintaining the processing ease of polymers. However, the hybrids have not contained enough MOFs to form continuous channels through the membrane, constraining the molecule’s transport to the polymer.
Berkeley Lab scientists fabricated a hybrid membrane in which MOFs account for 50% of its weight, about 20% more than other hybrid membranes, without undermining structural integrity.




