Electrode materials for lithium-ion batteries
N. Mughees | April 17, 2024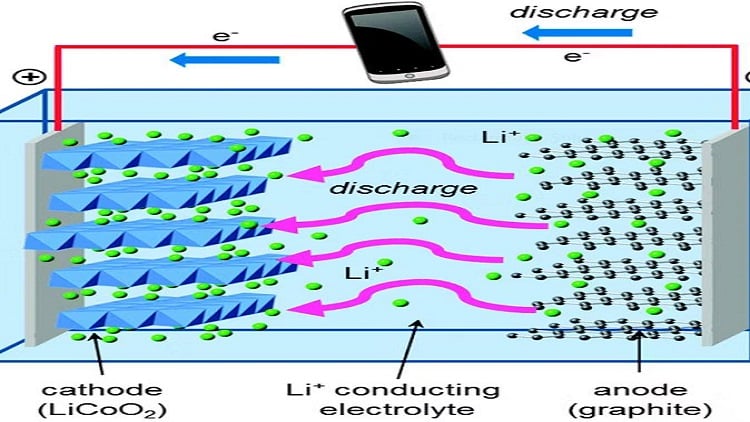 The anode gives off lithium ions to the cathode as the battery discharges, creating a current that flows from the anode to the cathode. As a result, the energy that powers the battery is released. Source: Islam and Fisher, Chemical Society Reviews, 2014, CC BY
The anode gives off lithium ions to the cathode as the battery discharges, creating a current that flows from the anode to the cathode. As a result, the energy that powers the battery is released. Source: Islam and Fisher, Chemical Society Reviews, 2014, CC BY
From electric vehicles (EVs) and grid storage to smartphones and laptops, lithium-ion batteries have transformed our world. It has two main parts: the anode and cathode electrodes. The anode gives off lithium ions to the cathode as the battery discharges, creating a current that flows from the anode to the cathode. As a result, the energy that powers the battery is released. When the device is plugged in, the cathode releases lithium ions and the anode receives them.
A lithium battery cell's cathode materials and metals can add 30% to 40% to the price tag, whereas anode materials usually make up around 10% to 15% of the overall cost. In this article, we will discuss the different types of materials that are being used as electrodes in lithium-ion batteries.
Anode materials
The ideal characteristics of an anode include high coulombic/electrical energy output, stability, strong conductivity and efficiency as a reducing agent. The following are some promising materials for lithium-ion batteries:
- Silicon: This abundant element has a theoretical capacity exceeding 3,500 mAh/g, but its large volume changes during cycling can cause electrode pulverization and capacity fade. Intensive research focuses on mitigating these challenges through nano structuring and composite designs.
- Metal oxides: Materials like tin oxide and lithium titanate offer good cyclability and safety but often fall short in terms of capacity. Tailoring their structures and compositions is crucial for unlocking their full potential.
- Graphite derivatives: Modified forms of graphite, such as hard carbon and graphene, offer improved capacity and cyclability compared to pristine graphite while maintaining its safety profile. Further research aims to optimize their pore structures and surface functionalities for even better performance.
Cathode materials
During discharge, the anode releases electrons into the battery, which are then taken up by the cathode, the other electrode. Our electronic gadgets get their power from these electrons as they go through an external circuit. Electric currents return to the cathode during charging, drawing lithium ions out of the electrolyte and reintroducing them to the cathode structure. Popular cathode materials include:
- Lithium cobalt oxide (LiCoO2): This classic material offers high energy density and good cyclability but suffers from high cost, limited abundance of cobalt, and safety concerns due to thermal instability.
- Lithium iron phosphate (LiFePO4): This phosphate-based material is much safer and more affordable than LiCoO2 but exhibits lower energy density. Doping and structural modifications are being explored to enhance its performance.
- Lithium nickel manganese cobalt oxide (LiNiMnCoO2, NMC): This family of materials offers a balance between energy density, cost and safety, making them popular choices for electric vehicles. Tailoring the nickel, manganese and cobalt ratios optimizes their properties for specific applications.
Other promising materials
In addition to the above-discussed popular electrode materials, researchers are looking into a number of other potential options:
- High-voltage spinel oxides: Materials like lithium nickel manganese oxide (LiNiMnO3) offer higher energy densities than NMCs but face challenges with cyclability and structural stability. Research focuses on improving their long-term performance.
- Lithium-rich layered oxides: These materials, like lithium nickel cobalt aluminum oxide (LiNiCoAlO2), boast even higher energy densities but require careful design to address voltage fade and stability issues.
- Beyond lithium: Sodium-ion batteries, with their abundant and low-cost sodium resource, are gaining traction as a potential alternative for grid storage applications. Research on cathode materials for these batteries is actively ongoing.
Recycled materials
Batteries may lose some of their effectiveness with time, but the precious metals and minerals used to make them are not really being wasted. One option to meet the increasing demand for lithium-ion batteries while keeping costs down is to incorporate more recycled content into the production of cathode and anode materials. Materials for recycling can originate from discarded products or from batteries that have reached the end of their useful life.
[See also: Efficiency in lithium recovery from spent batteries]
Recycling helps keep natural resources intact and minimizes negative effects on the environment by decreasing the demand for raw material extraction and mining. Greenhouse gas emissions can be reduced since it uses less energy compared to raw material extraction and processing. The amount of used batteries is growing but recycling them can reduce the amount of toxic trash that ends up in landfills. Moreover, there is a financial incentive for battery producers to embrace sustainable methods by using recycled materials instead of buying raw resources.
Conclusion
Perfect performance from lithium-ion batteries is only achievable with precise control over the amount of charge that can flow through the battery. Because of their fundamental importance, cathode and anode materials are required for a battery to function. Without them, the battery would not be able to operate. Moreover, one critical step toward the global electrification and clean energy goals is the incorporation of recycled content into the fabrication of cathode and anode materials of batteries.




