Metal under the microscope
Seth Price | September 17, 2022In middle school science class, most students look through a microscope at cells and thin sections of organic material. These microscopes use transmitted light to shine through a thin section. However, sometimes it is just as important to look at a magnified image of something thicker where light cannot pass through. In this case, some microscopes can be used to view reflected light that bounces off the surface of a sample.
Metallography is a field of study where metal samples are viewed under a microscope that uses reflected light in this manner. It is used to verify metallurgical processes are behaving properly, to look for defects and to verify microstructure. Microstructure is the shape, size and structure of the grains of metal as they are solidified from a melt or how they were altered during processing, such as deformation or heat treatment.
By itself, metallography is not a characterization technique. No metallurgist can look at an unknown sample and report its composition based on a micrograph alone. Instead, it can be used to back other tests, or decide if a process is working correctly on a known metal chemistry.
High-quality metallographic samples
To produce a high quality metallographic sample suitable for metallography, one must first ensure that the sample will not be chemically or structurally altered by the sampling process. This means samples should not be heated to the point of heat treating or losing the temper of the material and it should not be stressed beyond its yield point.
The sample must first be cut small enough to fit under the microscope. This should be done with either a low-speed saw or a saw that has a cooling system to prevent heat treating the surface. Once it has been sectioned, small samples can be mounted in epoxy or thermoset plastic for ease of handling. After that, it must be ground and polished with sandpaper or diamond wheels to a mirror finish. The purpose of the grinding and polishing is to remove scratches from the sectioning that may hide the grain structure. The sample is now mechanically ready for metallography.
If this sample with a mirrored surface were placed under a microscope, it would show up as a bright light, as the surface would reflect most of the light back through the eyepiece. To bring out the grain structure, the surface is lightly brushed with an etchant. When the sample was sectioned, the blade cut through the grains. The etchant attacks these incomplete grains and eats its way through to the next grain boundary. The art of metallography is etching just enough to remove the incomplete grains without attacking the grains underneath. This requires practice and experimentation with different etchants to find the right combination.
Applications in metallography
By itself, metallography should not be used for determining composition, but it can be used for verification purposes. It can be used to verify cold work, heat treatment and annealing and other such processes as well.
Brass mix-up
Suppose a supplier of brass bar stock has shipped cartridge brass for final products (70% Cu / 30% Zn) and free-machining brass (61.5% Cu / 35.5% Zn /3% Pb) for training purposes. When the brass arrives, it is unloaded, perhaps into the wrong storage facilities. On the surface, they both look like brass bars, but with metallography, the lead shows up as dark nodules in the brass.
 Cartridge brass, so called because it is used in ammunition cartridges; this particular image was taken from a .30-30 Winchester case near the headstamp. Source: Author’s image
Cartridge brass, so called because it is used in ammunition cartridges; this particular image was taken from a .30-30 Winchester case near the headstamp. Source: Author’s image
 Free machining brass from bar stock. The dark splotches are lead. Source: Author’s image
Free machining brass from bar stock. The dark splotches are lead. Source: Author’s image
Cast iron verification
When casting iron, the carbon content must be tightly controlled. If the carbon content is between 2.9% and 3.5%, the result is “gray” iron. From 3.5% to 3.8%, the result is “nodular” iron. Nodular iron is more ductile and is generally stronger than gray iron, which tends to fail in a brittle manner. This has everything to do with the microstructure.
In nodular iron, carbon tends to cluster and form graphite spheres in a ferrite matrix. When the material is stressed, any cracks that form must travel through the ferrite matrix and go around the graphite spheres, dispersing energy along the way.
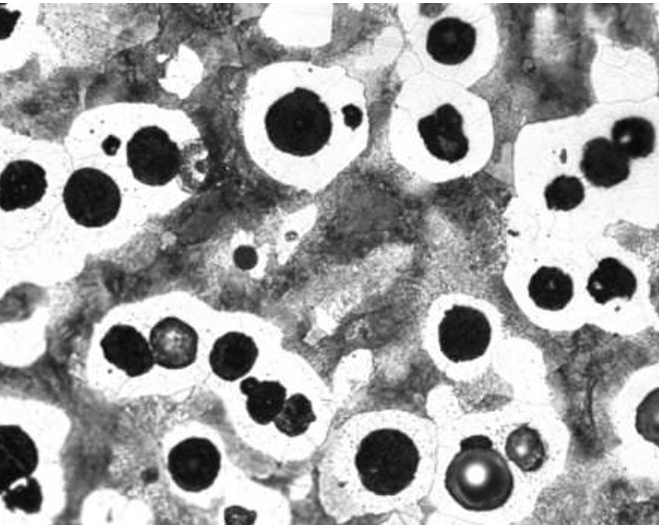 Nodular iron. The bottom left shows a graphite sphere protruding above the surface. Source: Author’s image
Nodular iron. The bottom left shows a graphite sphere protruding above the surface. Source: Author’s image
Gray iron, on the other hand, forms graphite flakes. Instead of cracks propagating through the ferrite matrix, they can form in these graphite flakes, which are much more brittle. Cracks end up traveling between brittle flakes, only having to pass through small sections of ferrite. This leads to a material that is much more brittle and has much lower tensile strength.
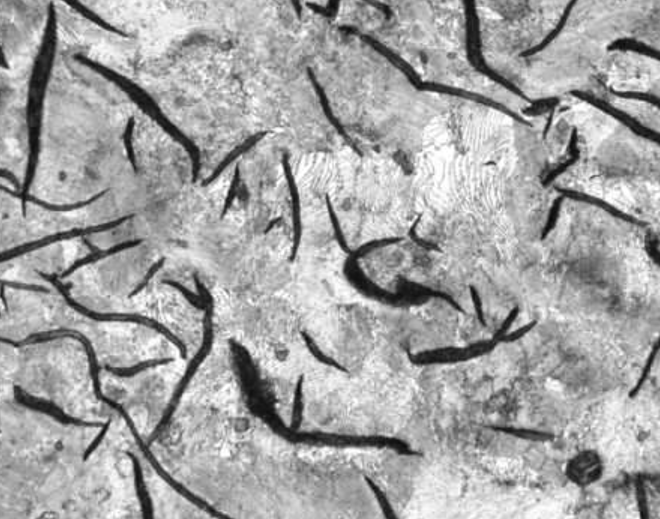 Gray iron: Notice the graphite flakes that form instead of graphite spheres. Source: Author’s image
Gray iron: Notice the graphite flakes that form instead of graphite spheres. Source: Author’s image
Iron foundries take samples of each heat and check for graphite flakes. Because there is such a marked difference between nodular and gray iron, they typically only perform a few polishing steps, as shallow scratches and debris cannot hide the microstructure.
Grain size and shape
Fine-grained metals, meaning metals with small grain sizes, are typically stronger and harder than coarse-grained materials. Sometimes, fine-grained materials lose ductility as well, as cracks can propagate along the grain boundaries where atomic bonds are the weakest. Also, if the deformation is not isostatic, such that the material is deformed in a certain direction (think rolling, pressing, drawing or extruding), the grains are deformed in this direction.
 Cartridge brass that has been rolled. Notice the grains are longer horizontally than vertically. Also, notice the scratches running nearly vertically; those are not the microstructure but a defect in metallography. Source: Author’s image
Cartridge brass that has been rolled. Notice the grains are longer horizontally than vertically. Also, notice the scratches running nearly vertically; those are not the microstructure but a defect in metallography. Source: Author’s image
When metals are cold-worked, (deformed below the recrystallization temperature, or below approximately 30% of the melting temperature), the grains are crushed. This means each cold-work operation makes the grain size smaller, and the material becomes a little less ductile.
Consider a brass wire drawing operation. Wire is made to a thinner gauge by pulling it through sizing dies, reducing its diameter. Each time the material passes through a die, it is cold-worked, decreasing its ductility. To restore the ductility, annealing steps (often an open flame) can be added along the path. The heat anneals, or allows the grains to grow again.
 The same cartridge brass after an appropriate heat treatment. Notice the grains are equiaxed (the same size in all directions). Source: Author's image
The same cartridge brass after an appropriate heat treatment. Notice the grains are equiaxed (the same size in all directions). Source: Author's image
Final thoughts
Ultimately, microstructure influences physical properties. The more information available from the microstructure, the more predictable the physical properties will be across a heat of molten metal or a lot of processed material. While it is not a characterization technique in itself, it can be used as a verification tool that backs data collected from other tests.
Image credit statement
These images were taken over the course of three separate undergraduate metallurgy laboratory classes at Virginia Tech in 2003-2005. The author processed some of these samples and took pictures on some of them. Because this was a class effort and they were commonly shared among classmates, the author would like to acknowledge the Materials Science and Engineering Department at Virginia Tech’s metallurgy classes from 2003-2005, who also contributed to the sample preparation and microscopy found in these images.




